Animal secretions (gross) often contain organiccarbon-based molecules that perform clever functions. Here are a few examples, accompanied by morsels of chemistry facts.
Hippo Sweat
 Hippos “sweat” a sunscreen that protects their nearly hairless skin from getting fried in the sun. This hippo sweat (which isn’t technically sweat) starts out clear but soon turns red and then brown on the hippo’s skin. The color – and the sunscreen properties – are mostly due to a couple of organic molecules that absorb ultraviolet wavelengths of light.
Hippos “sweat” a sunscreen that protects their nearly hairless skin from getting fried in the sun. This hippo sweat (which isn’t technically sweat) starts out clear but soon turns red and then brown on the hippo’s skin. The color – and the sunscreen properties – are mostly due to a couple of organic molecules that absorb ultraviolet wavelengths of light.
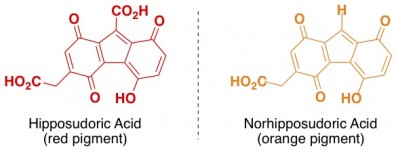
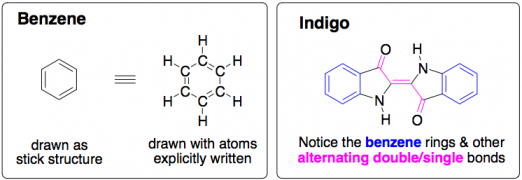
Chemistry Morsel: As a rule of thumb, molecules that contain alternating double and single bonds, such as benzene rings (below), are able to absorb UV light. When you start getting a lot of these alternating double/single bonds, it becomes likely that the molecule will also absorb visible light, which means it will appear colored.
Skunk Spray
When threatened, skunks spray a notoriously smelly concoction of chemicals consisting mostly of low molecular weightsmall, volatile thiols:
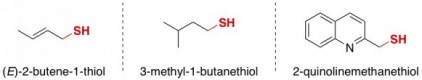
Chemistry Morsel: A thiolpronounced thigh-all (aka mercaptan) is a type of organic molecule that has a sulfur-containing part that looks like this: C–S–H (carbon bonded to sulfur bonded to hydrogen, colored red in the molecules above).

Sometimes chemists have to use thiols like this in lab, which usually results in disgruntled coworkers. In lab, we get rid of thiol stench (from gloves, countertops, glassware, etc.) using bleach. Bleach oxidizesin this case, makes sulfur-oxygen bonds the thiols, and the oxidized chemical products don’t really smell. Unfortunately, you can’t bleach your dog if he gets sprayed by a skunk.
Fire Ant Venom
When a fire ant stings a victim, it leaves behind a bit of venom. The main constituents of this venom are alkaloids such as piperidine.
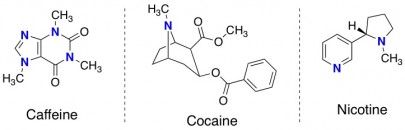
Chemistry Morsel. The term alkaloid is sort of a catch-all classification for small organic molecules that contain nitrogen, but don’t belong to another class of molecules such as amino acids. Some other famous alkaloids are caffeine, cocaine, and nicotine.