 Take a look at the ingredient list on the back of a Sour Patch Kids bag the next time you go to the movie theater, and you probably won’t be surprised to discover that this gummy candy contains a lot of chemicals. With such a complex ingredient list, you might be surprised to learn that the key feature of these snacks – their sourness – is actually quite simple. The sourness comes not from one chemical, not even one element, but from just one tiny little fraction of an atom.
Take a look at the ingredient list on the back of a Sour Patch Kids bag the next time you go to the movie theater, and you probably won’t be surprised to discover that this gummy candy contains a lot of chemicals. With such a complex ingredient list, you might be surprised to learn that the key feature of these snacks – their sourness – is actually quite simple. The sourness comes not from one chemical, not even one element, but from just one tiny little fraction of an atom.
Yup, protons are sour.
Protons are fractions of hydrogen atoms. When a hydrogen atom loses its electron, all that remains is a naked proton, often designated H+ because protons are positively charged.
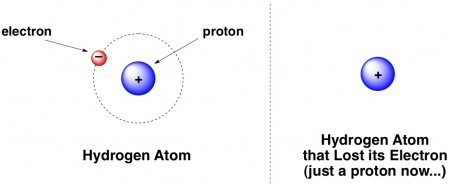
Obligatory chemistry joke that most people have probably heard:
A hydrogen atom walks into a bar. He says to the bartender, “I think I lost my electron.” The bartender asks, “are you sure?” The hydrogen says “I’m positive.”
Moving on.
The amount of naked hydrogen atoms – protons – running about in a mixture (such as in your candy-filled mouth) is directly related to the concept of acidity. By definition, when an acid is placed in a watery solution (such as in saliva), it increases the concentration of protons in that solution, usually because it contains a proton that can “fall off” into the water.*
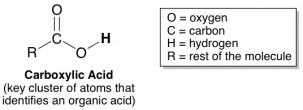
Our tongues detect high concentrations of protons as being sour. Thus, acids taste sour. There are tons of different types of acids out there, but fortunately, most acids that show up in food products are organic acids. Which means they’re fairly easy to recognize if you know their chemical structure. Organic acids are characterized by a cluster of atoms arranged just like this:
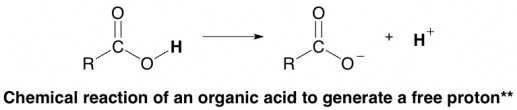
The hydrogen atom in that cluster is prone to falling off, a chemical reaction that can be written like this:**
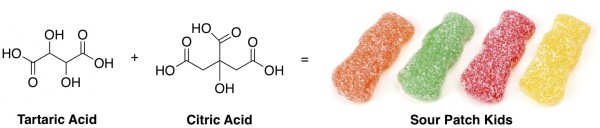
The acids in Sour Patch Kids that donate protons to your tongue are tartaric acid and citric acid, which look like this:
Another important acid for sour foods is lactic acid. This is the acid that is formed in the pickling process, and is responsible for the sour taste of sauerkraut and – of course – pickles.

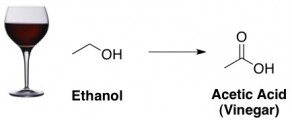
Finally, when wine goes sour it’s because alcohol (ethanol) has turned into vinegar (acetic acid):


5 Responses to Sour Patch Kids are Full of Protons