Retroviruses such as HIV are in some ways still a black box to scientists. The specific details of their mechanism of action are slowly being elucidated, but much is yet unclear. However, an article was published in Nature this past week sheds some light on how retroviruses work at the molecular level. The authors use X-ray crystallography, a technique that allows you to indirectly “see” atoms, to figure out how certain key molecules interact with each other. It’s an intriguing paper, but the subject matter is a bit dense and begs for a bit of background. So this week I’m going to do something a little different. At the end of the week I’ll post a summary of the article’s findings, but prior to that I’ll put up a couple of preparatory background posts.
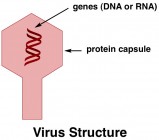 Today we’ll start with retroviruses. A retrovirus looks about the same as any other virus, but it functions significantly different that your run-of-the-mill influenza. HIV is the most significant retrovirus that infects humans; others include hepatitis B and human T-lymphotropic virus. Some retroviruses specifically infect animals and plants too; they are known for cats, cows, mice, monkeys, chicken, cauliflower, and more.
Today we’ll start with retroviruses. A retrovirus looks about the same as any other virus, but it functions significantly different that your run-of-the-mill influenza. HIV is the most significant retrovirus that infects humans; others include hepatitis B and human T-lymphotropic virus. Some retroviruses specifically infect animals and plants too; they are known for cats, cows, mice, monkeys, chicken, cauliflower, and more.
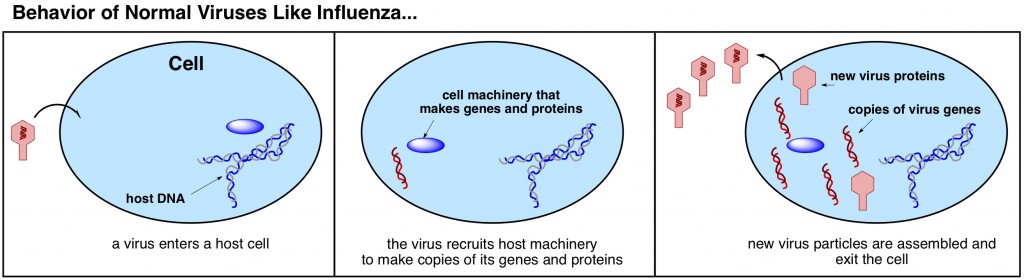
Viruses are essentially just genes, encapsulated in a protein. Most common viruses, such as influenza, function by entering a hostthe host is the organism, such as a person, that the virus has infected cell (such as a cell in your body) and hijacking the cell’s machinery to make copies of its own genes (below). Once encapsulated in new viral proteins, these gene copies are virus particles of their own right, and can exit the host cell to infect a new cell. This cycle can usually be halted by the host’s immune system; an effective immune system will eventually exorcise all of the virus particles – proteins, genes, and all – from the host’s body and cellular machinery can get back to performing its regularly scheduled functions.
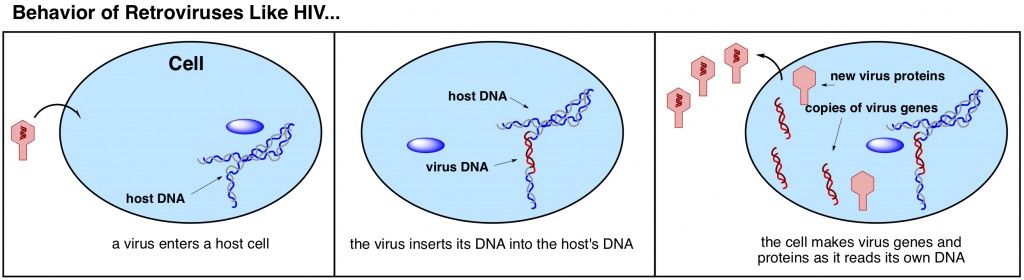
However, some viruses, called retroviruses, are particularly conniving. When they enter a host cell, they don’t call attention to themselves and demand that the host enzymes drop what they’re doing to come copy virus genes. Instead, they just casually snip open the host cell’s DNA, graft in their own genes, and seamlessly glue it all back together (below). The host’s own DNA now actually includes the code for making more virus particles. Thus, when the cell goes about its normal business of reading over its DNA code and making the prescribed protein precursors necessary for normal cell function, it comes across a section of its own DNA that tells it to produce virus proteins and genetic material. A good immune system may temporarily get rid of all the virus particles that are being churned out, but it can’t uninstall the bit of foreign virus DNA that has been integrated into host DNA.
This feature of retroviruses presents an excruciatingly difficult riddle for biologists to solve. How do you rid your body of a foreign organismwhether or not organism is the correct term for viruses, which are more like robots, is debatable when its DNA has become your own DNA? The riddle has no answer as of yet. Drugs that treat an HIV infection work to quiet the virus gene and prevent the cell from obeying its commands to make new virus particles. The drugs, however, don’t actually remove the virus DNA. Until very recently it was inevitable that the drugs would eventually be overpowered by virus.
Thus, many scientists are working intently at understanding exactly how retroviruses are able to sneak their DNA in. The more known about this enemy, the better to develop ways to fight it. A clear picture of the mechanism that retroviruses use to pull off this DNA integration stunt could aid in the development of new drugs to prevent and treat HIV infections.


One Response to The Machinery of Retroviruses Part 1: What is a Retrovirus?