(Part 1 here) X-ray crystallography is used by the authors of a recent Nature paper to visualize the structure of certain key molecules relevant to retroviruses. Though we usually think of X-rays as just a way of looking at our bones, these high energy light waves can also be used to “look at” atoms and molecules.
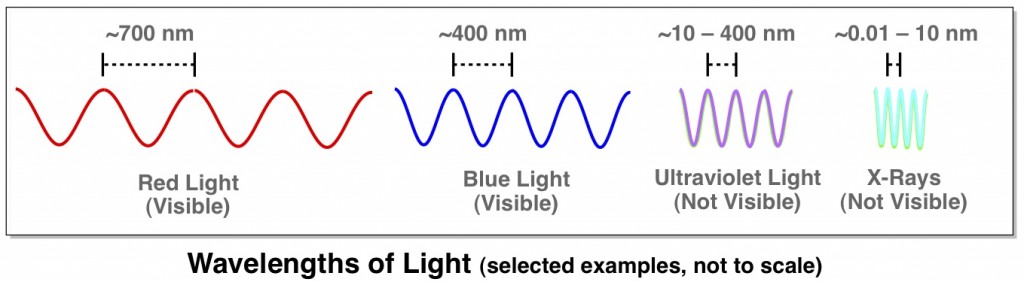
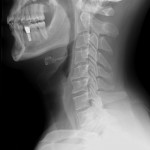 Like visible light, X-rays are a type of electromagnetic radiation. They have much shorter wavelengths though, and unlike visible light, the X-rays used for medical purposes can easily pass right through soft tissue like skin and muscles. This attribute of X-rays allows them to be used to take pictures of bones. To look at the bones in your neck, for example, a photographic plate is placed on one side of your neck and X-ray beams are shone through from the opposite side. The X-ray beams that pass through just tissue or air hit the photographic plate on the other side and darken it. The beams that run into bone, however, don’t make it through to the other side, and the photographic plate stays white where the beams would have hit.
Like visible light, X-rays are a type of electromagnetic radiation. They have much shorter wavelengths though, and unlike visible light, the X-rays used for medical purposes can easily pass right through soft tissue like skin and muscles. This attribute of X-rays allows them to be used to take pictures of bones. To look at the bones in your neck, for example, a photographic plate is placed on one side of your neck and X-ray beams are shone through from the opposite side. The X-ray beams that pass through just tissue or air hit the photographic plate on the other side and darken it. The beams that run into bone, however, don’t make it through to the other side, and the photographic plate stays white where the beams would have hit.
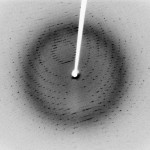
X-Ray Diffraction Pattern of Crystal
Things as tiny as molecules can also be visualized with X-ray beams, though the imaging technique is significantly different than that used in the doctor’s office. For the laboratory technique called X-ray crystallography, an X-ray beam is fired at a crystal of some molecule of interest. When the beam hits an atom, it bounces back (it is diffracted). The angle of the X-ray’s return depends on the position of the atom. By measuring the pattern of X-ray diffraction off a crystal, a skilled crystallographer can decipher or “solve” the structure of the crystal.
 For this technique, it’s very important that a nice tidy crystal is used (as opposed to say, a liquid, or an amorphous powder). In a crystal, all the individual molecules or small clusters of molecules are oriented exactly the same way. If the molecules are all facing different directions, the diffracted X-ray beams would point in random angles rather than making a clean cut pattern. Although it can be extremely tricky to decipher a diffraction pattern, the toughest part of getting an X-ray crystal structure is often the first step – growing a perfect crystal that is large enough to use for X-ray crystallography. Researchers can spend years trying to grow a crystal of complicated molecules like proteins.
For this technique, it’s very important that a nice tidy crystal is used (as opposed to say, a liquid, or an amorphous powder). In a crystal, all the individual molecules or small clusters of molecules are oriented exactly the same way. If the molecules are all facing different directions, the diffracted X-ray beams would point in random angles rather than making a clean cut pattern. Although it can be extremely tricky to decipher a diffraction pattern, the toughest part of getting an X-ray crystal structure is often the first step – growing a perfect crystal that is large enough to use for X-ray crystallography. Researchers can spend years trying to grow a crystal of complicated molecules like proteins.

