 Pharmaceutical companies sometimes get a bad rap, but most people don’t realize just how labor/money-intensive the process of drug discovery is. A recent JACSJournal of the American Chemical Society paper offers a little glimpse at the process – although this research was done by chemists at the University of Texas, not by a pharmaceutical company.
Pharmaceutical companies sometimes get a bad rap, but most people don’t realize just how labor/money-intensive the process of drug discovery is. A recent JACSJournal of the American Chemical Society paper offers a little glimpse at the process – although this research was done by chemists at the University of Texas, not by a pharmaceutical company.
The steps to develop a new pharmaceutical drug to treat a disease usually look something like this:
(1) Scientists do a bunch of research on a disease and identify one or more particular biological proteina receptor or enzyme in the human/animal body that seems to be related to the illness.
(2) Scientists set up an assay to screen zillionsok, hundreds or thousands of different organic molecules to see if they have any interaction with the biomolecule of interest.
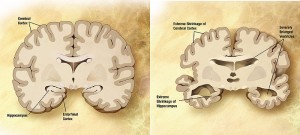 But sometimes a drug is needed for a disease that doesn’t have a clear biological protein target yet. This is the case with Alzheimer’s disease. There are currently several proteins that are suspected to play a role in Alzheimer’s, but targeting those biomolecules with drugs is tricky. Furthermore, this type of dementia seems to be due to multiple different abnormalities in brain cells, all going on simultaneously. For these reasons, it’s not too surprising that a silver bullet hasn’t been found yet to treat Alzheimer’s. (Photo credit: Wikimedia Commons.)
But sometimes a drug is needed for a disease that doesn’t have a clear biological protein target yet. This is the case with Alzheimer’s disease. There are currently several proteins that are suspected to play a role in Alzheimer’s, but targeting those biomolecules with drugs is tricky. Furthermore, this type of dementia seems to be due to multiple different abnormalities in brain cells, all going on simultaneously. For these reasons, it’s not too surprising that a silver bullet hasn’t been found yet to treat Alzheimer’s. (Photo credit: Wikimedia Commons.)
Because Alzheimer’s doesn’t have a clear biomolecule target, chemists at the University of Texas have taken a somewhat backwards approach to finding an Alzheimer’s drug. They call their approach atavistic.
Instead of choosing a protein to target with drug molecules, they gave mice a whole bunch of different organic molecules, and then checked to see if any of them had a positive effect on the mouse brains. Specifically, the researchers were looking to see if any of the would-be drug molecules caused neurogenesis – the growth of new brain cells – in the hippocampus.
 They were interested in neurogenesis because the growth of new brain cells seems likely to be correlated with a delay in the onset of Alzheimer’s. Older adults that exercise and participate in mentally engaging activities can stave off Alzheimer’s for a while. It’s tough to say for sure if sudoku or lifting weights stimulates neurogenesis, but a mouse study suggests this is the case. Mice that have toys and exercise wheels to play with have been shown to grow more new brain cells on a regular basis than mice in spartan environments.
They were interested in neurogenesis because the growth of new brain cells seems likely to be correlated with a delay in the onset of Alzheimer’s. Older adults that exercise and participate in mentally engaging activities can stave off Alzheimer’s for a while. It’s tough to say for sure if sudoku or lifting weights stimulates neurogenesis, but a mouse study suggests this is the case. Mice that have toys and exercise wheels to play with have been shown to grow more new brain cells on a regular basis than mice in spartan environments.
The researchers tested about 2000 different organic molecules, by giving mice ten at a time for a week. After a week, they looked at the mouse brains to see if new cells had grown in the hippocampus.
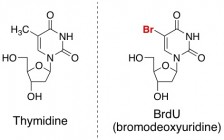
To tell new brain cells apart from old ones, the scientists gave the mice a tagged thymidine analog, called BrdU, along with the drug molecules. Thymidine is one of the building blocks of DNA. Since the mice were being given BrdU, this special version of thymidine would be incorporated into the DNA of new brain cells (but not old brain cells).
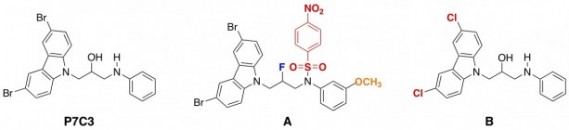
By this method, the scientists found one particular molecule, called P7C3, that seemed to cause a lot of brain cell growth. Next, they made a whole bunch of tiny alterations to the structure of the molecule, one change at a time, and tested each version of the molecule in the mice. P7C3 is depicted below, along with two of the modified versions that they also made and tested.
This type of experiment, which is almost always done when looking for drug candidates, is called a structure-activity relationship (SAR) study. It’s helpful for three main reasons:
(1) You might find a molecule that works even better than the original molecule.
(2) You can figure out structural changes that you can get away with without hurting the activity of the molecule. This could be useful if you wanted to later, say, attach a “tag” to the molecule that could help you track it down in the human body but wouldn’t affect the molecule’s behavior against a disease.
(3) You can input all the information you get about structural changes – what helps and what hurts in terms of desired activity – into a computer and have the computer come up with an idea of the shape/properties of the biomolecule target that your drug molecule could be interacting with. This could help you figure out what protein(s) are involved with the disease.
The researchers found that, for example, molecule A (above) worked better than the original P7C3, and molecule B didn’t work as well. They also made and tested around 60 other molecules with different structural changes – a LOT of work for a synthetic chemist.
Some active analogs seemed promising as potential drug candidates for Alzheimer’s – they didn’t seem to be toxic, could be taken as a pill rather than as an injection, etc. Furthermore, all the information the researchers gathered from their SAR study is expected to help with the identification of biomolecules involved with the disease.
Citation:
MacMillan, K., Naidoo, J., Liang, J., Melito, L., Williams, N., Morlock, L., Huntington, P., Estill, S., Longgood, J., Becker, G., McKnight, S., Pieper, A., De Brabander, J., & Ready, J. (2011). Development of Proneurogenic, Neuroprotective Small Molecules Journal of the American Chemical Society DOI: 10.1021/ja108211m


Pingback: ResearchBlogging.org News » Blog Archive » Editor’s Selections: Giraffes, Alzheimer drugs, the physics of gravity and the cold, cold winter
Pingback: RB Editor’s Selections: Giraffes, Alzheimer drugs, the physics of gravity and the cold, cold winter